UPDATE: Researchers at the Korea Research Institute of Bioscience and Biotechnology (KRIBB) have just announced a breakthrough that could revolutionize drug development. A newly developed human intestinal cell model allows for the precise detection of drug-induced gastrointestinal toxicity, a critical issue in preclinical drug testing.
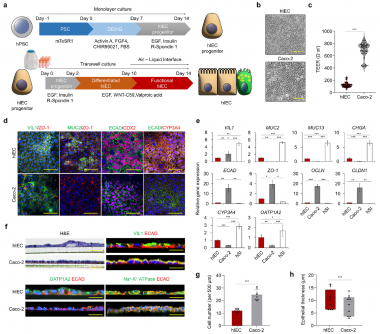
This innovative model, led by Dr. Mi-Young Son, closely mimics the structure and function of the human gut, enabling scientists to predict adverse effects with unprecedented accuracy. Conventional methods often fail to detect early signs of gastrointestinal damage, which can lead to costly failures in clinical trials. The new model boasts an impressive 94% overall accuracy in predicting gastrointestinal toxicity, with a remarkable 100% specificity for identifying safe drugs.
Gastrointestinal toxicity can manifest as severe side effects like vomiting, diarrhea, and mucositis—issues that frequently derail drug development programs. By recognizing these early warning signs, the hIEC model plays a crucial role in mitigating risks before drugs enter human trials.
Developed from human stem cells, the hIEC model replicates the cellular composition of the human intestine, including key cell types vital for nutrient absorption and mucus secretion. This advancement allows researchers to measure functional changes in the intestinal barrier, rather than merely assessing cell survival. The model demonstrated 92% sensitivity in identifying early intestinal barrier damage, detecting toxic signals from anticancer medications like paclitaxel that traditional tests often miss.
The study’s findings, published online on February 12, 2023, in Experimental & Molecular Medicine, indicate that drug-induced intestinal damage can now be predicted with high accuracy. The research team performed transcriptomic analyses, revealing that specific anticancer drugs can suppress essential genes tied to cell adhesion, hinting that barrier dysfunction can occur even before cell death.
Dr. Son emphasized the potential for this technology to transform personalized medicine, stating, “Our next goal is to expand this technology into a patient-specific organoid-based platform capable of precisely predicting intestinal toxicity.” These advancements could significantly reduce clinical trial failures and may even replace some animal testing methods.
The implications of this research extend beyond just drug development; they could lead to enhanced safety and efficacy in treatments for patients worldwide. With this new model, scientists can better understand how drugs interact with the human body, potentially saving lives by preventing harmful side effects from reaching patients.
As the medical community eagerly anticipates further developments from KRIBB, this breakthrough represents a pivotal moment in the intersection of biotechnology and patient care. For continuous updates on this emerging research, stay tuned as we follow the story.