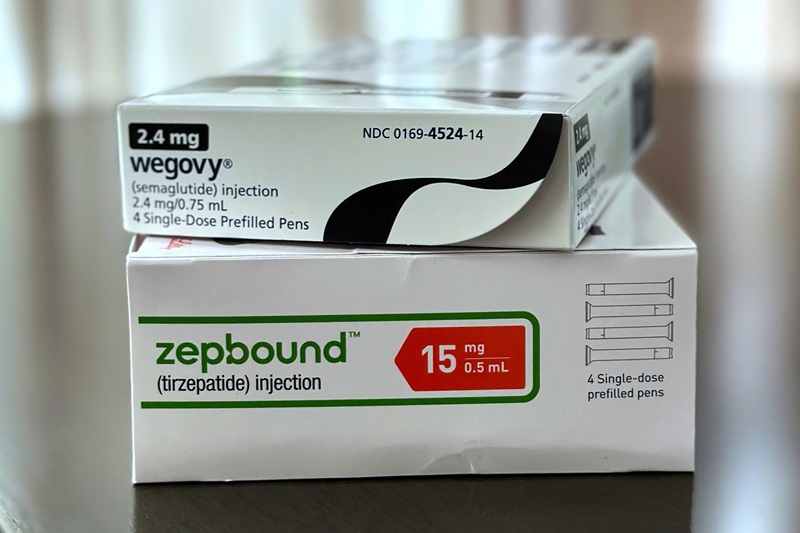
The demand for GLP-1 weight loss medications has surged in recent months, with approximately one in ten Americans reportedly using these drugs. As advertisements promoting these medications proliferate, concerns regarding their safety, costs, and accessibility have also emerged.
Dr. Jennifer McCauley, a specialist in weight management at UNC Health, stated, “They’re incredibly effective for weight loss.” The popularity of GLP-1 medications, which include well-known brands like Ozempic and Wegovy, is attributed to their ability to help users shed significant pounds quickly. However, the rise in usage has prompted a closer examination of potential side effects and the implications for the pharmaceutical industry.
Understanding GLP-1 Medications and Their Impact
GLP-1, or glucagon-like peptide-1, is a hormone that plays a crucial role in regulating appetite and insulin secretion. Medications that mimic this hormone have become increasingly popular as a solution for obesity. According to the U.S. Food and Drug Administration, these drugs can assist individuals in losing 5% to 10% of their body weight when combined with lifestyle changes.
Despite their effectiveness, the drugs are not without controversy. Reports indicate that prices can vary significantly, with some consumers facing costs exceeding $1,500 per month for out-of-pocket expenses. This creates a barrier for many potential users, especially those without insurance coverage that includes these treatments.
Furthermore, the National Institutes of Health has raised questions about the long-term effects of GLP-1 medications. While some individuals have experienced success, others have reported adverse reactions, including gastrointestinal issues. Dr. McCauley emphasized the importance of medical supervision when using these medications to mitigate risks.
Market Dynamics and Future Considerations
The pharmaceutical industry is closely monitoring the growing market for GLP-1 drugs. As demand escalates, companies are investing heavily in research and development to expand their offerings. This includes potential new formulations and combinations that could enhance effectiveness or reduce side effects.
However, as the market evolves, regulatory bodies are also increasing scrutiny. The U.S. Food and Drug Administration has begun to review the marketing practices surrounding these drugs, ensuring that advertisements accurately reflect both benefits and risks. Public health advocates are calling for transparent communication from pharmaceutical companies regarding potential side effects and the importance of professional medical guidance.
As the landscape of weight loss treatments continues to shift, both healthcare providers and patients must navigate the complexities of GLP-1 medications. While these drugs offer promising results for many, the associated costs and safety considerations remain critical factors in their widespread adoption.
The surge in GLP-1 usage reflects broader trends in health and wellness, highlighting the ongoing battle against obesity in the United States and beyond. As more individuals seek effective weight loss solutions, the dialogue around these medications will likely intensify, urging both consumers and healthcare systems to remain informed and vigilant.