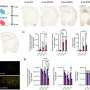
Research has unveiled the intricate processes behind the fermentation of ‘Nam Hom’ coconut cider, a beverage made from the naturally sweet coconut water known for its rich sugars and minerals. A study published on March 17, 2025, in Beverage Plant Research by a team led by Kriskamol Na Jom at Kasetsart University, explored how yeast selection influences the aroma, flavor, and nutritional value of this beverage.
Coconut water, while delicious, has a short shelf life, which limits its commercial appeal. Fermentation into cider not only extends its usability but also creates a low-alcohol beverage with desirable sensory characteristics. Yeast plays a crucial role in this fermentation, impacting sugar metabolism, alcohol production, and the formation of aromatic compounds. Different yeast strains produce distinct flavor profiles; some enhance fruity notes through ester production, while others maintain a cleaner taste that emphasizes the coconut’s natural qualities.
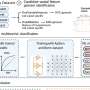
The study tracked the fermentation process using both physicochemical monitoring and extensive metabolomics and flavoromics analyses. By inoculating ‘Nam Hom’ coconut cider with two commercial yeast strains, K1-V1116 and EC-1118, researchers were able to examine the fermentation dynamics in detail. Employing multivariate statistical methods, the study identified three distinct fermentation stages: pre-fermentation, in-process, and final product.
During the fermentation, key metrics such as Brix and reducing sugars showed a steady decline, while the pH level decreased slightly, indicating an increase in acidity. Alcohol content rose significantly, reaching approximately 7–8%, a typical range for ciders, without evidence of lactic acid bacterial contamination.
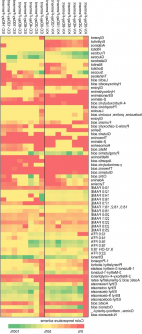
The analysis revealed that initial samples were rich in sugars like sucrose, glucose, and fructose. As fermentation progressed, primary amino compounds emerged, correlating with yeast activity. The final product was characterized by fruity volatiles, particularly esters like ethyl octanoate and ethyl 9-decanoate.
Progressive sugar depletion was observed, leading to the production of ethanol and other metabolites, along with an increase in glycerol, a common byproduct of yeast fermentation. The study noted stable levels of citric and lactic acids, with a slight rise in malic acid, which is indicative of a clean fermentation process.
Amino acids, particularly leucine and isoleucine, increased similarly in both yeast strains, linking to pyruvate-derived metabolism and the TCA cycle. Lipid analysis indicated a decrease in fatty acid methyl esters but an increase in free fatty acids, especially lauric and stearic acids, with higher levels noted in K1-V1116 fermentations.
Flavor profiling confirmed that esters were the dominant contributors to aroma. Yeast strain EC-1118 produced a more pronounced fruity profile due to its higher ethyl-ester content. The analysis also demonstrated strong correlations among sugars, ethanol, and esters, as well as between amino acids and flavor compounds. These findings reveal coordinated metabolic pathways that can be optimized to tailor the aroma and bioactive composition of coconut cider.
The study offers valuable insights for producers of coconut cider, emphasizing the importance of yeast selection and fermentation management. Both yeast strains showed promise, with K1-V1116 enhancing aroma through ester production, while EC-1118 facilitated robust fermentations with clear fruity notes. By strategically selecting yeasts and managing fermentation times, producers can create a range of flavor profiles, from dry and clean to rich and aromatic, all while preserving beneficial bioactive compounds.
This research contributes significantly to the understanding of coconut cider fermentation, potentially opening new avenues for value-added coconut beverages in the global market.