A collaborative team from the University of Manchester and the University of Oxford has successfully synthesized stable nitrogen chain radical anions under ambient conditions. This significant advancement, detailed in a study published on February 28, 2026, in the journal Nature Chemistry, marks a key development in the understanding of nitrogen chemistry.
Long-chain nitrogen ions, known as radicals, typically consist of chains of more than three nitrogen atoms. These molecules are commonly found in Earth’s ionosphere and other planetary atmospheres. Their high reactivity usually limits their stability to extreme conditions, such as ultrahigh pressure. The challenge of isolating nitrogen chains, particularly those containing more than three nitrogen atoms, has hindered scientific exploration due to their tendency to decompose rapidly. As they break down, they often release nitrogen gas (N2), making these compounds attractive for applications in propellants, explosives, and gas generators.
The authors of the study explained, “At ultrahigh pressures, the existence of different nitrogen phases has been demonstrated, and crystalline Nn (n > 3) chains identified in technologically relevant metal nitrides. Yet, studying nitrogen chain ions under ambient conditions presents a formidable challenge.” The possibility of uncovering new chemical properties and applications through a better grasp of these molecules is significant.
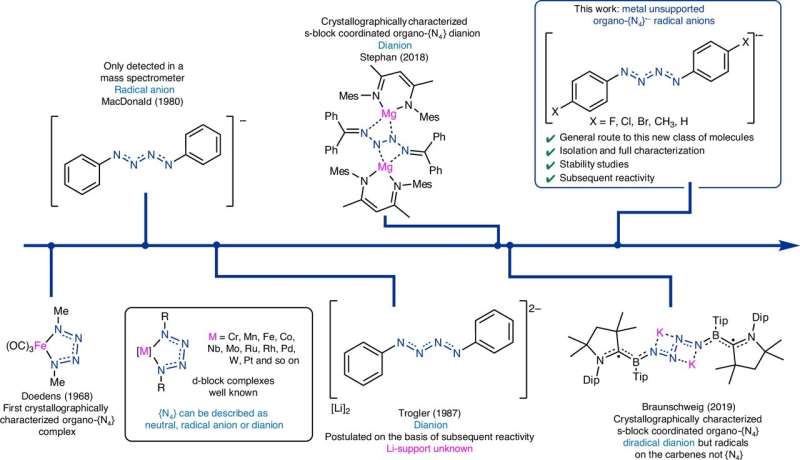
Historically, previous research efforts have attempted to isolate nitrogen chains in laboratory settings. Some studies have detected nitrogen chain ions using mass spectrometry or by trapping them at low temperatures, while others have synthesized metal-coordinated nitrogen chains. Unfortunately, the properties of these chains often changed due to the influence of the metal. While organic-substituted N4 chains have been documented, they have not been fully characterized or shown stability.
In the recent study, the research team reported the isolation of five crystalline molecules containing unsupported tetra-nitrogen radical anions ({N4}•−) at ambient conditions. This was achieved by reducing para-substituted phenyl azides with potassium graphite. Notably, one derivative, [(4-BrC6H4)2N4]•−, maintained its stability for over six weeks when stored under anaerobic conditions. The radical was found to be predominantly delocalized across the N4 chain, particularly at its terminal nitrogen atoms.
While most of the derivatives were stable, some exhibited instability and exploded upon isolation. The study also highlighted additional derivatives including [(4-FC6H4)2N4]•−, [(4-ClC6H4)2N4]•−, and [(4-MeC6H4)2N4]•−. The researchers observed variations in spin density across the {N4} chain and its stability based on aromatic substitution. Their computational and experimental electronic structure studies indicated that the {N4} chains exhibit partial multiple bond character, with significant radical character concentrated on the terminal nitrogen atoms bonded to the aromatic units.
Testing of stability and reactivity involved various reagents and conditions. These experiments determined that the N4 chain can cleave into N1 and N3 fragments, acting as a source of nitrene radical anions. Such characteristics suggest that these molecules could serve as storable nitrogen group transfer reagents. The team concluded that their findings align with previously conducted reactivity studies, demonstrating that the generation of a nitrene radical anion could lead to the production of an amide.
The implications of this research extend into industrial and scientific realms. The study authors state, “Efforts are now focused on exploring the additional reactivity patterns of {N4}•−-containing molecules, to enhance our understanding of their chemical properties and to fully unlock their potential as gram-scale storable nitrene synthons.” This breakthrough could pave the way for safer and more efficient applications of nitrogen in various sectors, providing accessible sources of reactive nitrogen species for chemical synthesis.
This article is based on research conducted by Reece Lister-Roberts and colleagues, published in Nature Chemistry, and underscores the importance of ongoing exploration in the field of nitrogen chemistry.