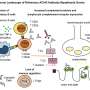
A recent study has identified a distinct immune signature associated with treatment-resistant myasthenia gravis (MG), a rare autoimmune disorder that significantly impairs muscle function. This breakthrough offers hope for improved patient outcomes by potentially guiding treatment strategies for those suffering from the severe form known as refractory MG.
Myasthenia gravis occurs when the immune system mistakenly attacks the neuromuscular junction, the critical connection between nerves and muscles. This leads to debilitating muscle weakness, affecting vital functions such as vision, movement, speech, swallowing, and even breathing. While many individuals respond well to current treatments, a subset of patients develops refractory MG, which does not adequately respond to conventional therapies.
Despite advances in medical science, there remains a significant challenge in predicting which patients will respond to treatment. As of now, there are no reliable biomarkers available, leaving healthcare professionals to make treatment decisions without clear predictive indicators.
Researchers from [Institution Name] conducted a comprehensive analysis, published on [Date of Publication], to identify potential biomarkers that could assist in anticipating treatment responses. Their findings highlight a specific immune profile that could distinguish between patients with treatable MG and those who may face a more resistant form of the disease.
The study analyzed blood samples from a diverse cohort of MG patients, employing advanced immunological techniques to assess immune responses. The researchers found that certain immune signatures were prevalent in patients with refractory MG, suggesting a link between these biomarkers and the severity of the disease.
Dr. [Researcher Name], the lead investigator, emphasized the importance of these discoveries. “Identifying these immune signatures is a critical step toward developing tailored treatment approaches for patients with myasthenia gravis,” they stated. “Our goal is to move beyond trial and error in treatment and provide patients with targeted therapies that can improve their quality of life.”
These findings could transform the management of myasthenia gravis by allowing for earlier identification of patients at risk of developing refractory MG. This proactive approach may lead to more effective treatment plans and improved patient outcomes.
As research continues in this area, the medical community remains hopeful that these insights will pave the way for advancements in the treatment of not only myasthenia gravis but also other autoimmune conditions that exhibit similar treatment resistance. The potential for personalized medicine in autoimmune diseases is an exciting frontier, and studies like this one are crucial in advancing our understanding and therapeutic options.
The study’s implications extend beyond individual patient care; they highlight the urgent need for further research into autoimmune diseases and the immune system’s role in their progression. As scientists and healthcare providers work together to unravel these complexities, patients with myasthenia gravis may soon benefit from more informed and effective treatment strategies.